|
In this case, the reaction is highly exothermic, and the drive towards a decrease in energy allows the reaction to occur. According to the drive towards higher entropy, the formation of water from hydrogen and oxygen is an unfavorable reaction. The entropy change for this reaction is highly negative because three gaseous molecules are being converted into two liquid molecules. Most real-world reactions are irreversible and result in an increase in the overall entropy of the universe because entropy is a measure of the dispersal of energy.\nonumber \].A reaction will be spontaneous if the ∆Suniverse is positive, and the reaction will not be spontaneous if the ∆Suniverse is negative.The ∆Suniverse is the sum of the entropy change of the system (∆Ssystem) and the entropy change of the surroundings (∆Ssurroundings).The entropy change of the universe (∆Suniverse) must be equal to or greater than zero for any process to be spontaneous based on the second law of thermodynamics.An increase in the number of gas molecules during a reaction often results in a positive entropy change.Dissolution can increase entropy, especially if a solution form is more disordered than the reactants. 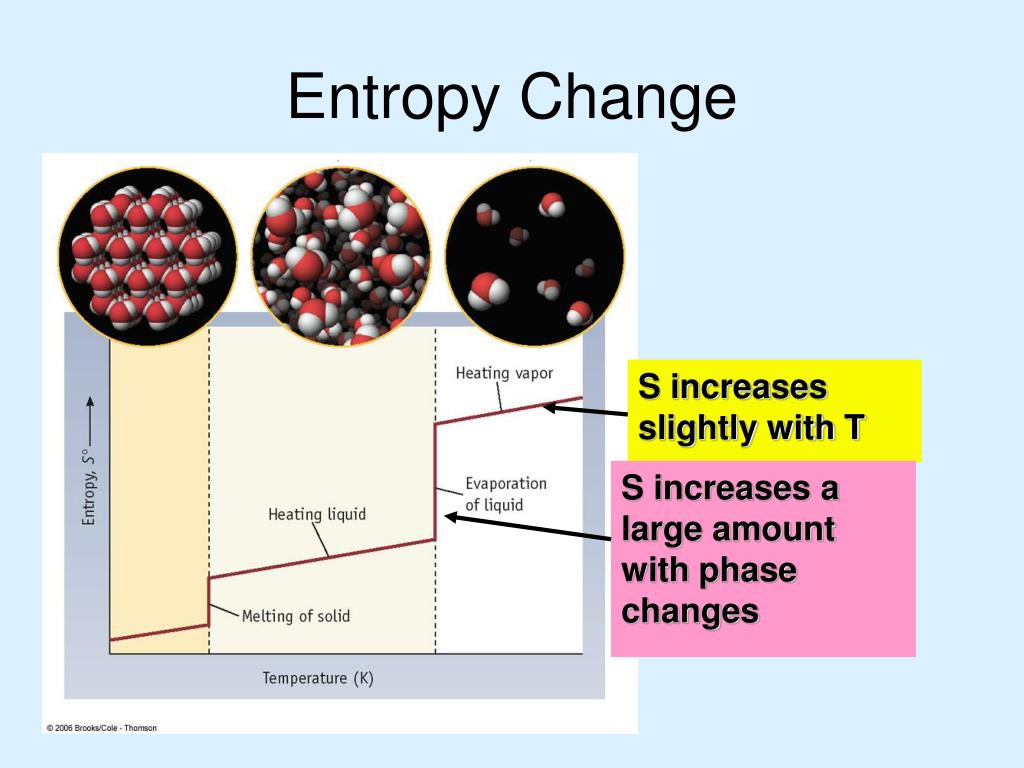
The entropy increases from solid to liquid and liquid to gas. Changes in physical states can also affect entropy. Figure 18.4.2 A Generalized Plot of Entropy versus Temperature for a Single Substance Absolute entropy increases steadily with increasing temperature until the melting point is reached, where it jumps suddenly as the substance undergoes a phase change from a highly ordered solid to a disordered liquid (S fus).Increasing the temperature normally increases the entropy of a system because it provides molecules with more energy to move.Entropy change can be calculated using the equation, ∆S = q_rev/T, where q_rev is the heat absorbed or released in a reversible process and T is the absolute temperature. The Third Law, then, gives us a method to find the absolute entropy for any substance: cool the substance to as close to absolute zero as possible and then.A negative entropy change (∆S A positive entropy change (∆S > 0) indicates an increase in randomness or disorder in the system and is associated with endothermic reactions.It’s calculated as the entropy of the final state minus the entropy of the initial state, ∆S = S(final) - S(initial).The entropy change (∆S) is the measure of the degree of randomness or disorder of a system from the initial to final state in a chemical reaction.Entropy is measured in joules per mole Kelvin (J/mol K) in the SI unit system. 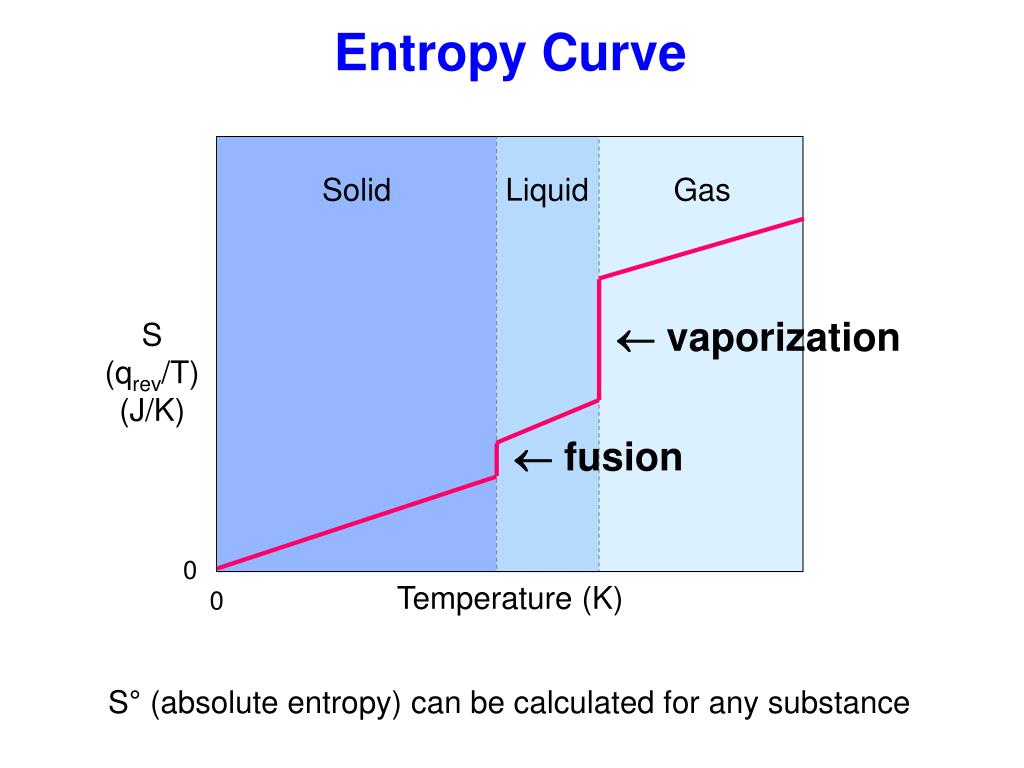
The more disordered a system is, the higher its entropy. This provides a reference point for calculating entropy changes.Įntropy values are always positive. The entropy of a pure crystalline substance at absolute zero is defined as zero in the third law of thermodynamics. It is a thermodynamic quantity, extensively used in chemical thermodynamics to predict whether a chemical process or reaction will occur spontaneously. It includes entropy changes occurring during phase transitions and reactions at standard conditions. Absolute entropy (S) refers to the entropy of a one mole of a substance at a standard state.Intramoelecular Force and Potential EnergyĪbsolute Entropy and Entropy Change Absolute Entropy and Entropy Change Absolute Entropy.Molecular and Ionic Compound Structures and Properties.Separation of Solutions and Mixtures Chromatography.Spectroscopy and the Electromagnetic Spectrum.Introduction to le Châtelier's Principle.Reaction Quotient and Equilibrium Constant.Reaction Quotietn and Le Châteleier's Principle. 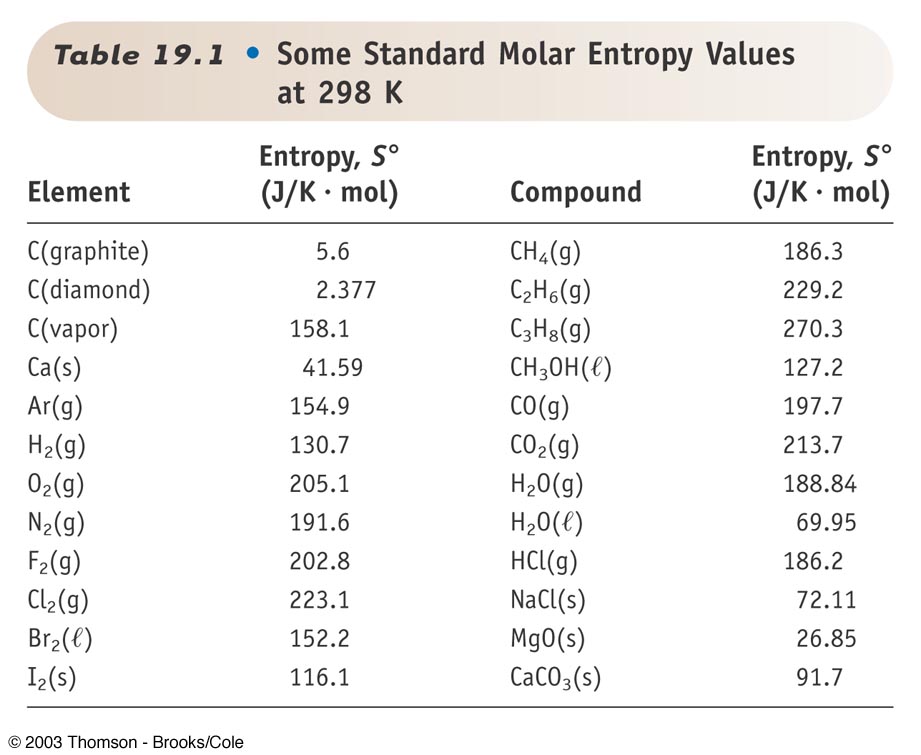
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
