
Therefore, photoelectrons arrive at the collector only if they leave the metal surface with enough kinetic energy to reach the collector. 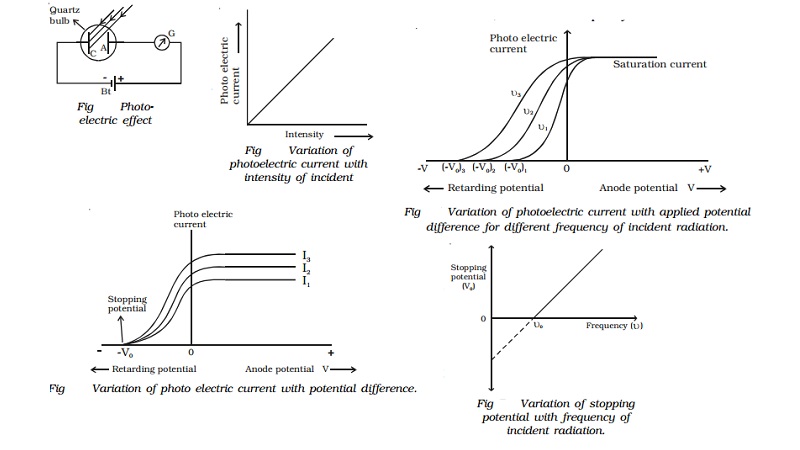
The electric force does work on the photoelectron. The electrons experienced an electric force directed away from the collector. When incoming photons hit the surface of the metal, photoelectrons were released. The metal collector was given a negative charge. The two were connected to a current and ammeter so that a circuit was made. Originally measured by Milikan by putting a metal collector at one end of an evacuated tube and an photoelectron emitting metal on the other. Since the frequency of a photon is directly proportional to its energy, as given by Planck's formula, the incident photons must have the minimum energy required to eject electrons. However, photoemission only occurs if the frequency of the incident photons is greater than or equal to the threshold frequency of the metal. This process takes very little time and all the energy needed to expel an electron is provided at once. Light energy arrives on the metal surface in discrete bundles, which are absorbed by atoms of the metal. Quantum physics: Albert Einstein solved the riddle of the photoelectric effect by applying Planck's quantum hypothesis. Experiments showed, however, that electron emission was essentially instantaneous: There was no measurable delay between the arrival of light on the metal surface and the emission of electrons. According to classical physics, it should take minutes to hours for a metal to emit electrons. The atoms would begin to vibrate and eventually absorb enough energy to eject an electron. E = Ek + W The energy of the photon equals the kinetic energy of the photoelectron plus the work required to release it from the metal.Ĭlassic physics: Perhaps the atoms on the surface of the metal absorbed the energy of the light waves. Ek = hf - W Ek = maximum kinetic energy of photoelectron, in J h = Planck's constant, in J f = frequency of incident photons, in Hz W = work function of metal, in J Which essentially shows the conservation of energy. The kinetic energy of the photoelectron is equal to the energy of the photon minus the work required to free the electron from the metal. If the frequency is high enough, the energy is high enough and a photoelectron is emitted) than the metal's work function, the photoelectron would leave with some kinetic energy. He also predicted that if an incident photon has more energy (high frequency, more energy. The work function, W, is related to threshold frequency, f0, by the equation: W = hf W = work function of a metal, in J h = Planck's constant, in J f = threshold frequency, in Hz Photons with a frequency greater than the threshold frequency have energy greater than the work function and electrons will be ejected. The work function describes the minimum energy that a photon can have to cause photoemission from metal. Related to Einstein's proposal that Planck's law relates to photon energy. Other metals require higher energy light such as x-rays, gamma etc. Alkali metals are commonly used since they require ultraviolet light only. Each type of metal has a characteristic threshold frequency. Depending on how much over the threshold frequency the incident light is, the emitted photoelectron can have a range of kinetic energies. 
The only thing that can effect energy of a photoelectron is the frequency of the light. The higher the photocurrent, the higher the stopping voltage. Above the threshold frequency, a photoelectric current is produced which is proportional to the intensity of light (number of photons). Any current would increase since more photons are released, showing that the number of photons is proportional to the number photoelectrons. If the threshold frequency is met, a higher intensity of light (increasing the number of incident photons) will increase the number of photoelectrons released but not their energy. The intensity of light does not effect the maximum kinetic energy of the released electrons. The intensity of the light does not matter does not effect photoemission if the frequency is not at or above the threshold frequency required for the metal. \)).Incident light must meet a minimum threshold frequency for photoemission to occur.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
